Levonorgestrel (Emergency Contraception)

- What is Levonorgestrel?
- Pharmacodynamics and Pharmacokinetics
- Contraindications
- Indications
- Side Effects
- Instructions for Use
- Interactions
- Special Instructions
What is Levonorgestrel?
Levonorgestrel is a synthetic progestogen that is widely used for birth control, natural «brothers» of which are responsible for normal pregnancy course. It changes cervical mucus secretion, makes this mucus more viscous and slows down sperm cells and egg movement.
However, negative feedback mechanism causes estrogen and progestogens production suppression that stops endometrial proliferative processes and leads to its involution. Thus, the agent reduces fertilization and embryo implantation probability.
Chemical Name
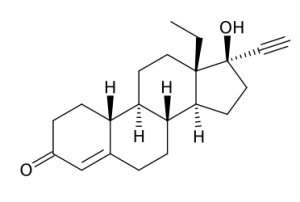 (8R, 9S, 10R, 13S, 14S, 17R)
- 13 - ethyl - 17 - ethynyl - 17 - hydroxy - 1, 2, 6, 7, 8, 9, 10, 11, 12, 13,
14, 15, 16, 17 - tetradecahydrocyclopenta[a] phenanthren - 3 - one
(8R, 9S, 10R, 13S, 14S, 17R)
- 13 - ethyl - 17 - ethynyl - 17 - hydroxy - 1, 2, 6, 7, 8, 9, 10, 11, 12, 13,
14, 15, 16, 17 - tetradecahydrocyclopenta[a] phenanthren - 3 - one
Release Form and Presentation:
- intrauterine system;
- micropyles;
- pills.
Chemical properties
Levonorgestrel is actively used as active hormonal contraceptives component.
Molecular hormone weight is 312,4 g per moll. This medicament is included in
oral contraceptives separately or in combination with other synthetic
hormones.
Pharmacodynamics and Pharmacokinetics
The drug causes structural changes in the endometrium, inhibiting its growth and thereby reducing implantation possibility. It also increases cervical mucus viscosity, which prevents sperm cells penetration into the uterus, and reduces the menstrual bleeding amount. If the drug is a part of a therapeutic intrauterine system, it has a direct local impact on endometrium growth, fallopian tubes and cervical canal mucus.
Preparations based on levonorgestrel have high 100% bioavailability. After penetration, the drug is completely and quickly absorbed through gastrointestinal tract walls and enters systemic blood circulation.
A single dose of 0.75 mg provides the maximal concentration in blood after 1 – 2,3 hours. This synthetic hormone has a high degree of binding to plasma proteins – about 55% (albumin and globulin).
Levonorgestrel is metabolized in the liver, resulting in forming pharmacologically inactive metabolites. Half-life period is about one day. Drug clearance appears in the form of inactive metabolites with urine and slightly – with feces.
After IUS installation hormone recovery rate into uterine cavity is initially 20 mcg per day, during 5 years it is reduced to about 11 mcg per day. The therapeutic intrauterine system can be used for women who receive hormonal replacement therapy in combination with oral or transdermal estrogens without a gestagen component.
However, if the egg has fixed, levonorgestrel emergency contraception can’t change anything: for the above reasons it does not have an abortive effect and is most effective when unprotected sexual intercourse occurred before ovulation. That is why this agent has no negative effects on the fetus but has a dangerous side effect: ectopic pregnancy risk caused by slow egg movement through fallopian tubes.
Contraindications
Absolute contraindications for Levonorgestrel:
- hypersensitivity;
- severe liver or bile passages diseases (ingestion);
- post-biliousness period;
- adolescense;
- pregnancy;
- lactation period.
Additional contraindications:
- recurrent or acute inflammatory pelvic organs diseases;
- cervicitis;
- endometritis after labor;
- urogenital system infections;
- septic abortion carried out during the last 3 months;
- dysplasia or uterine cervix or uterus malignoma;
- tumor dependant on progestogen (breast cancer);
- uterine bleeding of unknown origin;
- uterine development abnormalities acquired or congenital (uterine cavity deformity, fibromyoma);
- tumors or other severe liver diseases.
Indications
- emergency postcoital contraception for women after unprotected sexual intercourse;
- intrauterine therapeutic system for long-term contraception in idiopathic menorrhagia;
- IUS for endometrial hyperplasia prevention during estrogen replacement therapy.
Side Effects
During clinical tests the following adverse reactions of synthetic hormones were detected:
- sickness, fatigue, lower abdominal pains;
- headaches, heavy or poor menstrual bleeding, menstrual disorders;
- dizziness, breast swelling, and tenderness;
- diarrhea, vomiting.
IUS side effects:
- irritability, bad mood, headaches;
- migraines, emotional instability, sickness;
- acne, skin rashes, eczema and itching, alopecia, urticaria fever;
- excessive flatulence, distension, abdominal pains;
- uterine bleeding, spotting;
- amenorrhea, menstrual disorders, ovarian benign tumors (cysts);
- dysmenorrhoea, lower abdominal pains, cervicitis;
- vulvovaginitis, breast swelling, and tenderness;
- weight gain, backaches, edema;
- decreased sexual desire, endometritis, uterine perforation.
Instructions for Use
Depending on pharmaceutical form dosage regimen is completely different. The drug is administered orally or as a part of an intrauterine device.
Instructions for emergency contraception
Orally. levonorgestrel is taken in pills within 96 hours after unprotected sexual
intercourse. Average dose ranges from 0,75 to 1,5 mg.
The substance can be used in a single dose. It is not recommended to take this
drug for emergency contraception more than 1 time in 4 – 6 months.
Instructions for IUS
IUS installation procedure is carried out under doctor’s supervision. The
device is inserted into the uterus and stays there for up to 5 years. Periodic
examinations of patient’s condition are necessary.
Interactions
- Levonorgestrel in combination with rifampicin, primidone, phenytoin, and barbiturates has a lower contraceptive effect because of steroid metabolism process acceleration;
- In combination with ampicillin, chloramphenicol, phenoxymethylpenicillin, sulfamethoxypyridazine, nitrofurantoin, neomycin may enhance breakthrough bleeding. This happens because of sex steroids enterohepatic cycle suppression due to intestinal flora changes;
- The drug reduces the effectiveness of anticoagulant for oral preparation, antispasmodic, antihyperglycemic, and antihypertensive drugs;
- The drug impedes diazepam and chlordiazepoxide metabolism processes, which may lead to these substances accumulation in blood plasma.
Special Instructions
- In patients with heavy weight, the drug has less pharmacological activity.
- When implanting substance under the skin to patients with severe liver diseases, liver function monitoring must be carried out every 8 – 12 weeks.
- If within 6 weeks of therapy background menstruation hasn’t come (6 weeks from previous one), it is necessary to define and exclude pregnancy.
- In case of recurrent prolonged and heavy blood-tinged discharges between menstruation periods, it is necessary to conduct additional tests to confirm or correct diagnosis.


