What is Advair Diskus?
Advair Diskus is a prescription medication for COPD and asthma treatment. The drug produces anti-inflammatory and bronchodilator effects.
[ithshop code=”advair” tpl=”short” page_id=”2189″ dummy=”txt”]
This is a combination of two active substances: Fluticasone and Salmeterol. Fluticasone is a steroid that inhibits the release of substances in the organism that cause inflammation. Salmeterol is a bronchodilator and works by relaxing the airways muscles to improve breathing process.
- In people with COPD, this medication is used for long-term treatment;
- In people with asthma, the drug is used for short-term treatment as far as asthma symptoms are well controlled with some other drugs.
My Canadian Pharmacy sells the following dosages of Advair Diskus: 25mcg / 50mcg, 25mcg / 125mcg, 25mcg / 250mcg.
Release form: inhalation powder.
Pharmacologic Effect
The active substances Salmeterol and Fluticasone have different mechanisms of action. Salmeterol prevents bronchospasm, Fluticasone improves the lung function and prevents exacerbations. Advair Diskus can be an alternative for patients who take both an agonist β2-adrenergic receptors and inhaled corticosteroids.
-
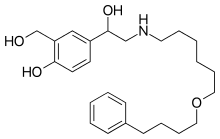 Salmeterol is a selective adrenergic β2-agonist of long-term action (up to 12 hours), which has a long side chain which binds to an external domain of the receptor. The pharmacological properties of Salmeterol offer protection against histamine-induced bronchoconstriction and long-lasting bronchodilation (lasting at least 12 hours) than the β2-adrenergic receptor short-acting agonists. The onset of bronchodilator effect occurs within 10-20 minutes.Salmeterol is a strong and long-acting inhibitor which releases mast cells of the pulmonary tissue such as histamine, leukotriene and prostaglandin D2.
Salmeterol is a selective adrenergic β2-agonist of long-term action (up to 12 hours), which has a long side chain which binds to an external domain of the receptor. The pharmacological properties of Salmeterol offer protection against histamine-induced bronchoconstriction and long-lasting bronchodilation (lasting at least 12 hours) than the β2-adrenergic receptor short-acting agonists. The onset of bronchodilator effect occurs within 10-20 minutes.Salmeterol is a strong and long-acting inhibitor which releases mast cells of the pulmonary tissue such as histamine, leukotriene and prostaglandin D2.
Salmeterol inhibits the early and late phases of response to inhaled allergens; the latter lasts over 30 hours after a single dose, i.e. when bronchodilator effect is over. A single administration of Salmeterol attenuates bronchial hyperreactivity of the bronchial tree. This indicates that Salmeterol in addition to the effect of bronchodilator has an additional effect, which clinical significance is not completely established. Advair mechanism of action differs from the anti-inflammatory effect of glucocorticoids. At therapeutic doses, Salmeterol has no effect on the cardiovascular system. -
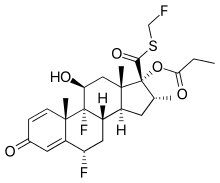 Fluticasone is corticosteroid for local application. When inhaled in the recommended doses, Fluticasone has a pronounced anti-inflammatory and anti-allergic action in the lungs, which reduces the clinical symptoms and exacerbations of the diseases accompanied by airway obstruction. The substance restores the patient’s response to bronchodilators reducing the frequency of their application. Fluticasone, inhaled at the recommended doses, is not accompanied by side effects typical of systemic corticosteroids. With prolonged use of inhaled Fluticasone at recommended doses, the maximum daily secretion of adrenal hormones remains in the normal level in both adults and children. After the transfer of patients treated with other inhaled corticosteroids to Fluticasone, the daily secretion of adrenal hormones is gradually returning to normal, despite the previous and current periodic use of oral corticosteroids. This indicates a recovery of adrenal function in the background of inhaled Fluticasone use. With prolonged use of Fluticasone, the backup function of the adrenal cortex also remains at the normal level, as evidenced by an increase in the normal production of cortisol in response to appropriate stimulation (please note that the reduction in adrenal reserve, caused by the previous therapy, can be maintained for a long time).A study of 318 adult patients with persistent asthma showed that when using double Fluticasone dose for 14 days, there is a slight increase in the frequency of adverse events associated with the action of beta-adrenergic agonist (tremor – 1 patient [1%] 0 patients at usual dosage; palpitations – 6 patients [3%] 1 patient [<1%] at the usual dose, convulsions 6 patients [3%] 1 patient [<1%] at ordinary dose). At the same time the frequency of adverse events associated with the inhaled corticosteroid action is stagnant (eg, oral candidiasis – 6 patients [6%] of 16 patients [8%] at the usual dose, hoarseness – 2 patients [2% ], 4 patients [2%] at the usual dose) compared to the conventional treatment scheme (1 inhalation 2 times / day). Thus, a double dose of the medicine can be used in cases when patients require additional short (14 days) course of corticosteroid therapy.
Fluticasone is corticosteroid for local application. When inhaled in the recommended doses, Fluticasone has a pronounced anti-inflammatory and anti-allergic action in the lungs, which reduces the clinical symptoms and exacerbations of the diseases accompanied by airway obstruction. The substance restores the patient’s response to bronchodilators reducing the frequency of their application. Fluticasone, inhaled at the recommended doses, is not accompanied by side effects typical of systemic corticosteroids. With prolonged use of inhaled Fluticasone at recommended doses, the maximum daily secretion of adrenal hormones remains in the normal level in both adults and children. After the transfer of patients treated with other inhaled corticosteroids to Fluticasone, the daily secretion of adrenal hormones is gradually returning to normal, despite the previous and current periodic use of oral corticosteroids. This indicates a recovery of adrenal function in the background of inhaled Fluticasone use. With prolonged use of Fluticasone, the backup function of the adrenal cortex also remains at the normal level, as evidenced by an increase in the normal production of cortisol in response to appropriate stimulation (please note that the reduction in adrenal reserve, caused by the previous therapy, can be maintained for a long time).A study of 318 adult patients with persistent asthma showed that when using double Fluticasone dose for 14 days, there is a slight increase in the frequency of adverse events associated with the action of beta-adrenergic agonist (tremor – 1 patient [1%] 0 patients at usual dosage; palpitations – 6 patients [3%] 1 patient [<1%] at the usual dose, convulsions 6 patients [3%] 1 patient [<1%] at ordinary dose). At the same time the frequency of adverse events associated with the inhaled corticosteroid action is stagnant (eg, oral candidiasis – 6 patients [6%] of 16 patients [8%] at the usual dose, hoarseness – 2 patients [2% ], 4 patients [2%] at the usual dose) compared to the conventional treatment scheme (1 inhalation 2 times / day). Thus, a double dose of the medicine can be used in cases when patients require additional short (14 days) course of corticosteroid therapy.

Pharmacokinetics
The simultaneous inhalation of Salmeterol and Fluticasone does NOT affect the pharmacokinetics of each of these substances.
Salmeterol
Salmeterol acts locally in the lung tissue and, therefore, its content in the plasma is not correlated with the therapeutic effect.
The data on the pharmacokinetics of Salmeterol is limited because it is technically difficult to determine very low concentrations of drug in plasma after Advair Diskus therapeutic doses inhalation.
Fluticasone
Absorption and distribution
After Fluticasone inhalation the absolute bioavailability in healthy people makes 10-30%. In patients with asthma and chronic obstructive pulmonary disease (COPD) there observed a lower concentration of Fluticasone in plasma. Systemic absorption occurs primarily in the lungs, it is more rapid in the beginning, but then slows down.
A part of the inhaled Fluticasone dose may be swallowed but the systemic action is minimal due to the low solubility of Fluticasone in water and intensive metabolism in the “first pass” through the liver. The bioavailability of Fluticasone in its absorption from the gastrointestinal tract is less than 1%.
As the inhalation dose increases, there observed a linear increase in the concentration of Fluticasone in plasma.
Fluticasone has a relatively high degree of binding to plasma proteins (91%).
Metabolism and clearance
Fluticasone is rapidly eliminated from the blood, mainly as a result of metabolism involving CYP3A4 isozyme with forming inactive carboxyl metabolite.
Fluticasone has a high plasma clearance (1150 ml/min). The final T1 / 2 is about 8 hours.
Renal clearance of Fluticasone is insignificant (<0.2%).
Fluticasone is excreted through the intestine mainly in the form of a hydroxylated metabolite.
Indications for Use
 The drug is intended for the treatment of asthma in patients who are prescribed the therapy with beta2-agonists and long-acting inhaled corticosteroids:
The drug is intended for the treatment of asthma in patients who are prescribed the therapy with beta2-agonists and long-acting inhaled corticosteroids:
- Patients with poor control of the disease against the background of continuing monotherapy with inhaled corticosteroids with periodical use of short-term beta2-agonists;
- Patients with adequate control of the disease during therapy with inhaled corticosteroids and long-term beta2-agonists;
- As a starting maintenance therapy in patients with persistent asthma (daily occurrence of asthma symptoms, daily use of means for the quick relief of symptoms) in the presence of indications for corticosteroids to achieve control of the disease.
The maintenance therapy in patients with COPD and FEV1 < 60% of the predicted values (up to inhaled bronchodilator) and repeated exacerbations when the disease severe symptoms persist despite the regular bronchodilator therapy.
Mode of Application
Canadian Advair is intended for inhalation use. It is recommended to use the medicine regularly, even in case of asthma attacks absence. In the treatment of reversible obstructive diseases of the respiratory system, patients should use consistent scheme and regular monitoring of their condition. With each reception, it’s necessary to review the prescribed dose and, if possible, reduce it (you should choose the lowest effective dose). It is forbidden to change the dose without consulting a doctor.
- For inhalation, open and charge the plastic device changing the position of the lever;
- Grapple the mouthpiece bow with lips;
- Do inhalation;
- Close the device.
* The indicator of doses shows the number of doses remaining.
The duration of use and a normal dosage should be determined by a physician. At the beginning of therapy, patients usually use the medicine twice a day to control asthma symptoms. After reaching the total control of asthma symptoms, the medication dosage is reduced to 1 dose per day. The amount of Fluticasone in the preparation is determined by a physician, taking into account the patient’s condition and tolerance of Advair Diskus for asthma.